 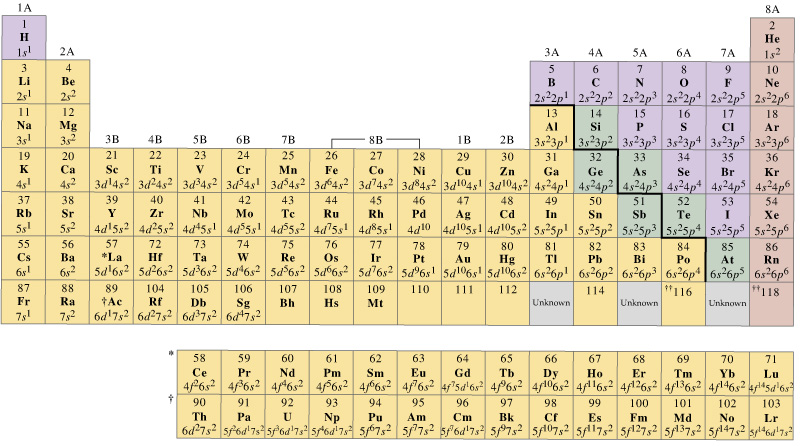
As you will see, this is reflected in important similarities in the chemical reactivity and the bonding for the elements in each column. Within each column, each element has the same valence electron configuration-for example, ns 1 (group 1) or ns 2 np 1 (group 13). 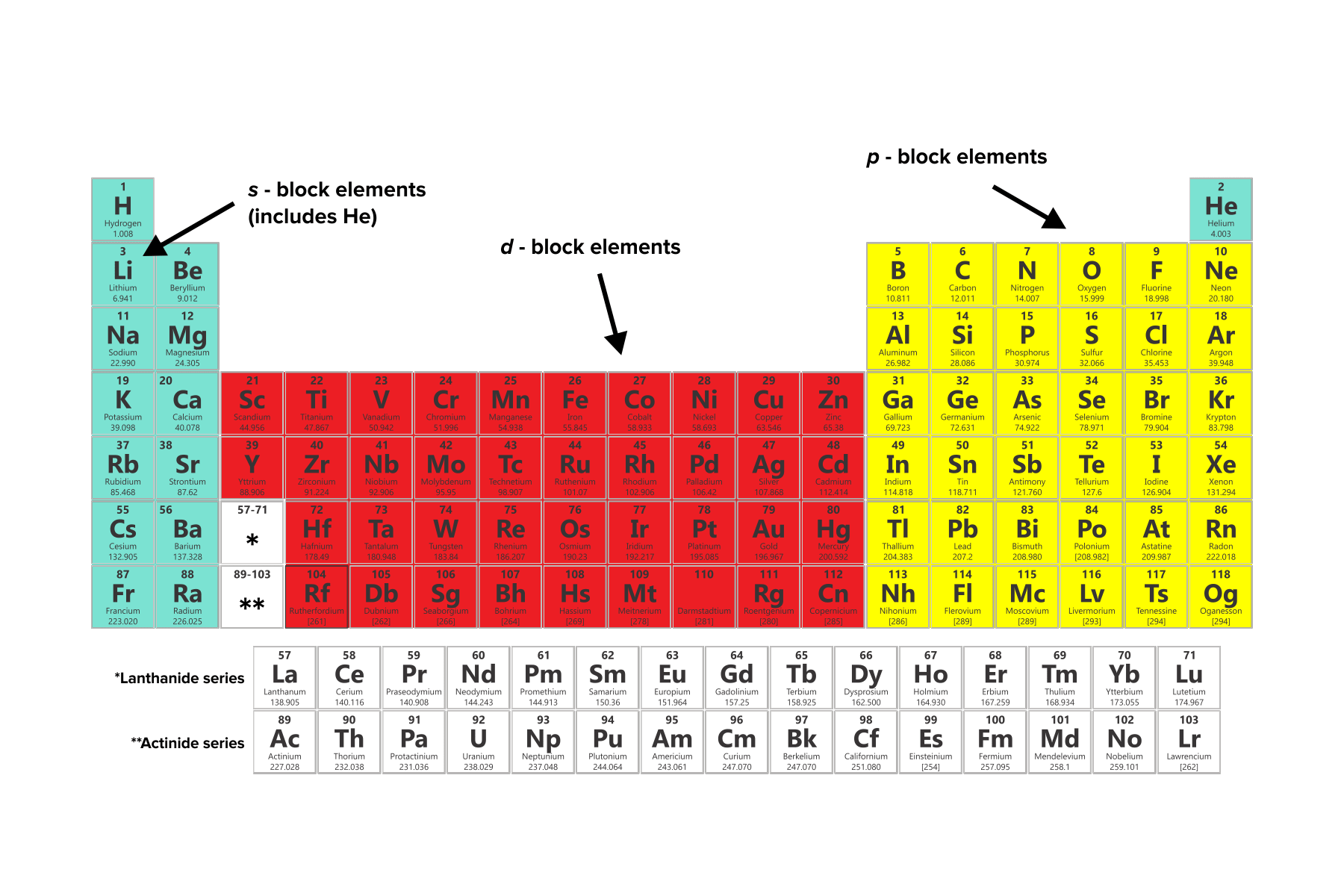
At the bottom lie the 14 columns of the f block, elements in which the ( n − 2) f orbitals are filled.īecause two electrons can be accommodated per orbital, the number of columns in each block is the same as the maximum electron capacity of the subshell: 2 for ns, 6 for np, 10 for ( n − 1) d, and 14 for ( n − 2) f. In between are the 10 columns of the d block, elements in which the ( n − 1) d orbitals are filled. The six columns on the right, elements in which the np orbitals are being filled, constitute the p block. As a result, the periodic table can be divided into “blocks” corresponding to the type of subshell that is being filled, as illustrated in Figure 2.3.1.įor example, the two columns on the left, known as the s block, consist of elements in which the ns orbitals are being filled. Although the table was originally organized on the basis of physical and chemical similarities between the elements within groups, these similarities are ultimately attributable to orbital energy levels and the Pauli principle, which cause the individual subshells to be filled in a particular order. Elements in the same Group have the same number of valence shell electrons.\)Īs you have learned, the electron configurations of the elements explain the otherwise peculiar shape of the periodic table.(valence shell electrons, the number of electrons in the highest energy level, shown in red) The electrons in the valence shell (highest energy level) are given in red.Ĭan you see a pattern, or trend, in the electronic configuration of the atoms in each group and in each period? The electronic configuration in shell notation is given for an atom of each of the elements. The table below for the main group elements is set out just like the Periodic Table of the elements. No ads = no money for us = no free stuff for you! Trends in the Number of Valence Electrons ⚛ Going down a Group of the Periodic Table from top to bottom, the energy of the valence shell electrons increases. ⚛ The valence shell electrons of elements in the same Period of the Periodic Table occupy the same energy level (shell). ⚛ Going across a period of the Periodic Table from left to right the number of valence electrons increases. ⚛ Elements in the same Group of the Periodic Table have the same number of valence shell electrons (electrons in the highest energy level). Trends in number of valence electrons (electrons in the highest energy level):. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
